|
| |
|
Cell Stacks |
|
|
|
Current Density |
MMW |
Neutral Plates |
Number of Plates |
Plate Configurations |
Voltage & Amperage
|
It has been
brought to my attention that confusion exists, among experimenters,
about what a Stack of cell plates is and when and why multiple stacks are used. I
will attempt to explain it for you and provide some pros and cons for
using them.
|
|
I will be analyzing cell stack efficiency
and explaining the reasons for additional
neutral plates, additional
stacks, and what they accomplish. Cell Stacks is another topic that no one seems to
explain; yet most companies incorporate multiple stacks into their
cells. For what it is worth, the following is My opinion; my inside view of the process.
What is a cell stack?
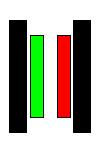
Basically, a water "Cell Stack" is a group of electrodes consisting of a positive and a
negative, and any number of neutral electrodes between them.
For instance ( + - ) consists of a positive electrode and a
negative electrode. Together, they create a single water compartment
between them. We call that a "Cell". ( + n - ) consists of one neutral between positive
and negative. One neutral creates 2 water compartments between positive
and negative. ( + n n n - ) consists of 3 neutrals between positive and
negative. 3 neutrals create 4 water compartments between positive and
negative. Each water compartment is considered a "Cell" and a group of
cells is considered a "Cell Stack".

The most popular cell configuration for 12 volt systems is 7
plates in Series. It consists of 5 neutral plates separated by a
single positive and a single negative electrode (a cell stack). Its
configuration looks like this ( + n n n n n - ). The 7 plates create 6
water compartments between the positive and the negative plates; count
them. Each of those water compartments creates a Voltage Drop that can
be individually measured. That voltage drop will be equal to the voltage
source divided by the number of water compartments. If 12 vdc is applied
to positive and negative, the voltage dropped across each water
compartment will be 2 volts; do the math --- 12 vdc divided by 6 water
compartments =
2.vdc. 7 plates in series is capable of producing 1 LPM @ 16 amps. The
late great Michael Faraday tells us that each water compartment can
produce 10.44 ML PM of HHO, per amp. 10.44 x 6 = 62.64 x 16 = 1002.24
ML PM.
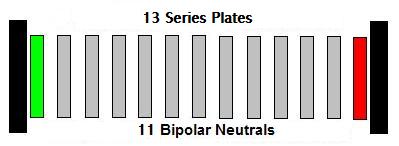
The most popular cell configuration for 24 volt systems is 13
plates in Series. It consists of 11 neutral plates separated by a
single positive and a single negative electrode (a stack). Its
configuration looks like this ( + n n n n n n n n n n n - ). The 13
plates create 12 water compartments between the positive and the
negative plates; count them. Each of those water compartments creates a
Voltage Drop that can be individually measured. That voltage drop will
be equal to the voltage source divided by the number of water
compartments. If 24 vdc is applied to positive and negative, the voltage
dropped across each water compartment will be 2 volts; do the math ---
24 vdc divided by 12 water compartments = 2.vdc. 13 plates in series is capable of
producing 1 LPM @ 8 amps. The late great Michael Faraday tells us that
each water compartment can produce 10.44 ML PM of HHO, per amp. 10.44
x 12 = 125.28 x 8 = 1002.24 ML PM.
Have you noticed that higher voltage requires more plates and uses
less amperage to produce more HHO gas?
|
Multiple
Cell Stacks
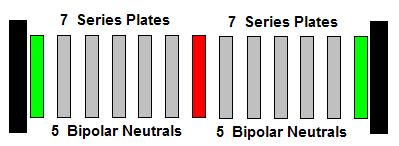
Generally speaking, we recognize a group of electrodes as a "Cell". We
usually do not include the word "Stack". I think that is what causes the
confusion when we have multiple groups of Cells sharing the same
positive or negative electrode. For instance:
- 2 Cell Stacks would look like this ( + - + ). Each cell stack is
sharing the same negative electrode.
- 2 Cell Stacks could also look like this ( - n n + n n - ). Each
cell stack is sharing the same positive electrode. Or (+ n n - n n +
). Each cell stack is sharing the same negative electrode.
- 3 Cell Stacks could look like this (+ - + - ) or this ( - n n n +
n n n - n n n + ). Each cell stack is sharing either a positive or a
negative electrode.
|
Why use Multiple Cell Stacks?
This is where it gets complicated. In the examples above, I stated that
the most popular 12 vdc system uses 7 plates in Series. It is popular
because it is proven to be a very efficient configuration. It is capable
of producing 1 LPM @ 16 amps; compared to a 6 plate cell stack that
would only produce 835 MLPM @ 16 amps. Plus, 7 plates lowers plate
voltage to around 2 vdc, where as 6 plates would lower voltage to around
2.4 vdc. The lower 2 volt plate voltage allows the cell stack to operate
continuously for longer periods of time than the 6 plates 2.40 vdc,
because it produces less heat; about 40% less. If it produces 40% less
heat, then it can operate 40% longer --- while producing 1 LPM.
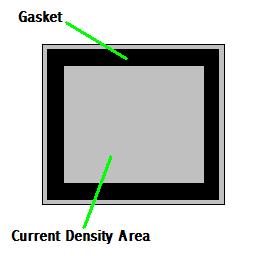
The catch to this is the size of the plates. The plates require
enough Active Surface Area to distribute 16 amps without causing
overheating. The late great Michael Faraday tells us that 1 square inch
of surface area can distribute 0.54 amps. According to my calculations,
16 amps would require about 30 sq. inches. That equates to a surface of
about 5.5 x 5.5 square. That is the size requirement "between" the plate
gasket, not the size of the plate. To accomplish this, the plates would
need to be 6 to 6.5 inches square.
Now tell me, how many cells have you seen advertised with 4 inch
plates? Cells that can produce 1 LPM or more? How can they do it, when 7
plates is about the maximum that can reasonably be used without
introducing excess heat? The answer is "Multiple Cell Stacks". The
smaller the plates are, the more stacks you will needed to create 1 LPM.
Here are some examples, but keep in mind, the size mentioned is the
Active Surface area inside the gasket; the area the water touches.
Gaskets are generally 1/4 to 1/2 inch in diameter, so a 1/2 inch gasket
will require a plate about 1 inch wider:
- 5.5 x 5.5 inch plates only need 1 cell stack ( + n n n n n - )
to produce 1 LPM @ 16 amps
[Current Density is 16.34 amps per cell stack]
- 3.9 x 3.9 need two cell stacks ( + n n n n n - n n n n n + ) to
produce 1 LPM @ 16 amps.
[Current Density is 8.21 amps for each cell stack].
- 3.5 x 3.5 need two cell stacks ( + n n n n n - n n n n n + ) to
produce 829 MLPM @ 16 amps, and 3 cell stacks to produce 1.244 LPM @
almost 20 amps ( + n n n n n - n n n n n + n n n n n -)
[Current Density is 6.62 amps for each cell stack]
- 3 x 3 need two cells stacks ( + n n n n n - n n n n n + ) to
produce 609 MLPM @ 16 amps, and 3 cell stacks to produce 913 MLPM @
almost 15 amps ( + n n n n n - n n n n n + n n n n n -), and 4 cell
stacks @ nearly 20 amps to produce 1.218 LPM
( + n n n n n - n n n n n + n n n n n - n n n n n +)
[Current Density is 4.86 amps per cell stack]
- 2.5 x 2.5 need two cells stacks ( + n n n n n - n n n n n + ) to
produce 423 MLPM @ nearly 7 amps, 3 cell stacks to produce 635 MLPM @
just over 10 amps, and 5 cell stacks to produce 1.059 LPM @ almost 17
amps.
[Current Density is 3.38 amps per cell stack]
Heat Efficiency
All of the above are efficient configurations for 12 volt systems that
can consistently produce HHO while producing minimal amounts of heat.
They all can accomplish the same efficiency as long as their Current
carrying capacity is not exceeded. But do multiple cell stacks heat the
water faster? The answer is yes and no. Lets say one stack is producing
1 LPM @ 16 amps and we add a second stack that also produces 1 LPM @ 16
amps. Both cell stacks are heating the water they share, so yes the
water will heat twice as fast. But a cell stack that produces 2 LPM @ 32
amps will create the same amount of heat as 2 stacks that use 16 amps
each. Perhaps someone smarter than me will create a calculator for
predicting the length of time a cells stack can operate without
overheating the water.
Almost all HHO
Companies over estimate the maximum amperage of their cell stacks
because they don't know how to properly calculate it. For
more details on Current Density , read my article.
|
Improving MMW
Some companies use MMW to calculate the efficiency of their cell stacks.
MMW is the milliliters produced per watt of power, in a minutes time.
Volts time Amps = Watts. I have
already shown you that adding more plates in series increases the HHO
production without increasing the amperage. The MMW of the 7 plate
Series cell, using 12 volts and 16 amps, has a base MMW of 5.208; not
accounting for temperature variations or loses. By adding one more plate
in series, MMW would increase to 6.088. It does so because LPM increases
by 167 MLPM while voltage and amperage remain the same. But you will find
that adding that 8th plate in series increases the resistance between
positive and negative and makes it harder for electrolysis to produce
the gas. The electrolyte mix concentration must be increased in order
to achieve that same amperage. The plate voltage decreases to 1.71 vdc
because another water compartment has formed. If you are using KOH or
NaOH electrolyte, their minimum operating voltage, between the plates, is
1.67 and 1.69 volts respectively. That is barely enough electrical
pressure for electrolysis to take place. That is going to cause a very
slow warm up period. It may take hours for the cell to create enough
heat to conduct well enough to pass 16 amps. That is very very heat
efficient, but it does not make good quantities of HHO on Startup. We
have found that maintaining 2 volts or close to 2 vdc for plate voltage
is the best for long operating times along with good HHO production. As
plate voltage exceeds 2.25 volts, cell operating time decreases. That is why
a 7 plate cell operates 40% longer than a 6 plate cell.
The MMW of a cell stack is affected by voltage changes. Some
companies lower the Operating Voltage from 12 volts and 10 amps, to 10
volts and 10 amps. They then calculate the MMW and tell you it is over
unity. Leave their web site. They are trying to scam you into believing
their product has superior MMW efficiency. You are not going to operate
your cell at 10 volts.
The MMW of a cell stack is also affected by temperature changes in the gas
output. As temperature increases, MMW efficiency decreases. The
temperature of the gas is directly affected by the temperature of the
plates, which affects the temperature of the water. And what most
companies that use MMW as the measuring stick do not realize, is that
MMW decreases, the longer the Cell Stack operates. Current Density has more influence on heat than plate voltage. I
encourage you to study my articles on
MMW and
Current Density. Conclusion
HHO Generators come in various shapes and sizes. Most companies
started their business by building a single small Cell Stack, using
small electrode plates; they fit into smaller places. As demand for more HHO increased, they started adding
multiple individual Cell Stacks in order to make more gas. Someone
eventually came up with the idea of combining Cell Stacks; by sharing
common electrodes and sharing the same water and bubbler. Most companies still use a single
plate size and design. It lowers their cost of production. They only
need one size of plates, and one size of gaskets. They only have to
change the length of the bolts and sometimes the size and shape of the
end plates. So before you invest in an HHO Generator, make sure the
Cell's dimensions
will fit your application. If you need a lot of HHO gas, use as few Cell
Stacks as you reasonably can. 6x6 inch plates can handle about 16 amps.
7 plates in Series can produce 1 LPM @ 16 amps; and do it efficiently.
4x4 inch plates needs 4 Cell Stacks of 7 plates in series in order to
make 1 LPM @ 16 amps. 4x6 inch plates need 2 Cell Stacks of 7 plates in
Series to produce 1 LPM @ 16 amps. Don't forget we calculate current
density by the size of the space inside the plate gasket. Something to
think about.
I hope this article helps you. I wrote it specifically to point out
short comings of most HHO companies out there. Some don't want to tell
you any details about their Cell Stacks; some will tell you anything to
make a Sale; very few will tell you everything.
I figure that if I educate the buyer, they will ask the companies for
more details, and perhaps the companies will clean up their acts. And,
there is always the chance that a Company will read this information
and learn something.
|
|
Comments can be sent to:
mailto:david@hho4free.com |
|
|
|
|
|
|
|
|
|
|
|
|